|
Lide, (ed.), CRC Handbook of Chemistry and Physics 1999-2000 : A Ready-Reference Book of Chemical and Physical Data (CRC Handbook of Chemistry and Physics, CRC Press, Boca Raton, Florida, USA, 79th edition, 1998. I am grateful to Professor Pekka Pyykkö (University of Helsinki, Finland) who provided the nuclear quadrupole moment data in this and the following two references. Where given, data for certain radioactive nuclei are from this reference. Oxygen, we can see from our periodic table of elements, it has a molar mass of 16.00 grams per mole. And then last but not least, we have oxygen here. Hydrogen has a molar mass of 1.008 grams per mole, 008 grams per mole. Mason in Multinuclear NMR, Plenum Press, New York, USA, 1987. So carbon has a molar mass of 12.01 grams per mole and now we can think about hydrogen in the same way. I am grateful to Professor Robin Harris (University of Durham, UK) who provided much of the NMR data, which are copyright 1996 IUPAC, adapted from his contribution contained within this reference. 5, John Wiley & Sons, Chichester, UK, 1996. Harris in Encyclopedia of Nuclear Magnetic Resonance, D.M. Magnetogyric ratio, γ (10 7 rad T ‑1 s -1) Table of NMR-active nucleus propeties of tungsten Ĭommon reference compound: Na 2WO 4/D 2O. Kuchitsu in Quantities, Units and Symbols in Physical Chemistry, Blackwell Scientific Publications, Oxford, UK, 1988. Interactive periodic table with up-to-date element property data collected from authoritative sources. Masses, nuclear spins, and magnetic moments: I.When using the unit g/mol, the numerical value of the molar mass of a molecule is the same as its average mass in. The molar mass has units of g/mol or kg/mol. A mole of substance is equal to Avogadros number (6.023×10 23) of that substance. For further information about radioisotopes see Jonghwa Chang's (Korea Atomic Energy Research Institute) Table of the Nuclides The molar mass is the mass of one mole of substance, whether the substance is an element or a compound.Naturally occurring isotope abundances: Commission on Atomic Weights and Isotopic Abundances report for the International Union of Pure and Applied Chemistry in Isotopic Compositions of the Elements 1989, Pure and Applied Chemistry, 1998, 70, 217.This table gives information about some radiosotopes of tungsten, their masses, their half-lives, their modes of decay, their nuclear spins, and their nuclear magnetic moments. This site explains how to find molar mass.Further data for naturally occuring isotopes of tungsten are listed above. The reason is that the molar mass of the substance affects the conversion. 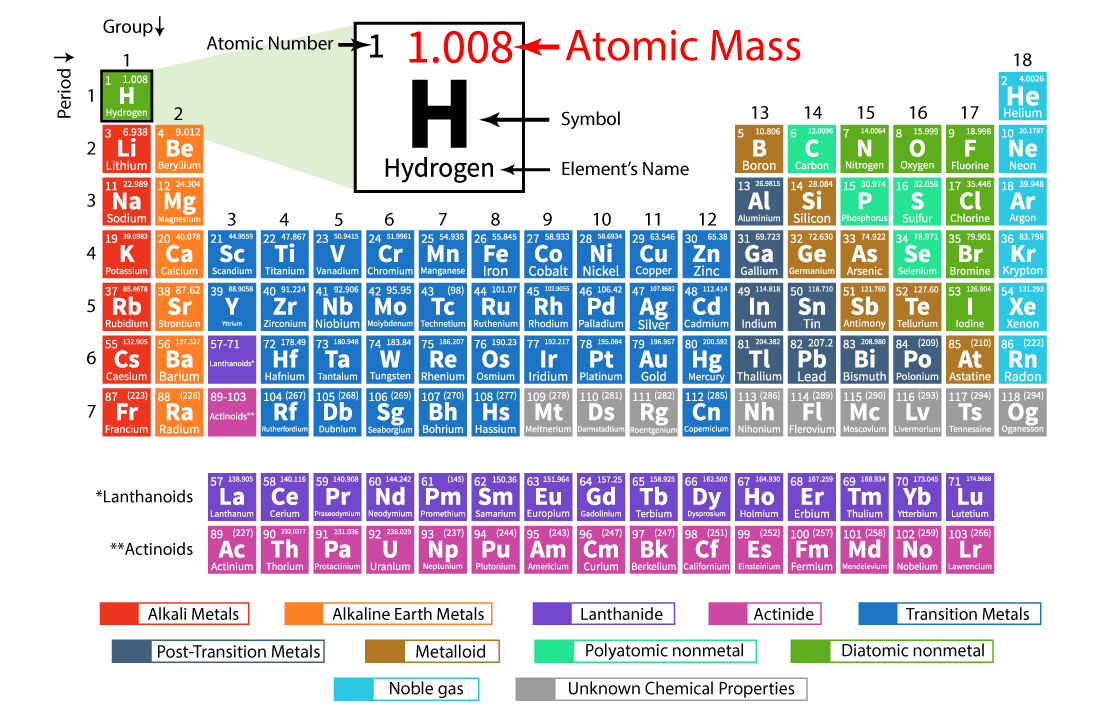
To complete this calculation, you have to know what substance you are trying to convert. These relative weights computed from the chemical equation are sometimes called equation weights.Ī common request on this site is to convert grams to moles. For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) 28.02 amu. /chemical-periodic-table-of-elements-with-color-cells-vector-illustration-840464844-ff61af51081b4fb1959504105e232e01.jpg)
For a molecule (for example, nitrogen, N 2) the mass of molecule is the sum of the atomic masses of the two nitrogen atoms. The formula weight is simply the weight in atomic mass units of all the atoms in a given formula.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. The concept of molar mass can also be applied to compounds. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.įinding molar mass starts with units of grams per mole (g/mol). For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. Using the periodic table, the molar mass of Potassium (K) is 39.10 g/mol. The molar mass is expressed in grams per mole (g/mol). 
To determine the molar mass of an element, you can refer to the periodic table. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The molar mass of an element is the average mass of one mole of atoms of that element. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100. If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
